With a sensitivity one thousand times higher than CAGE, nanoCAGE presents powerful new possibilities for the analysis of small-sized samples. Using nanoCAGE, as few as 1,000 cells is enough to detect capped RNA transcripts in a cell and map the genomic regions that cause a gene to be expressed, providing a detailed picture of how gene expression is regulated. nanoCAGE thus overcomes the difficulty of obtaining large quantity of cells for studying and future diagnosing diseases such as cancer.
CAGEscan complements nanoCAGE by pinpointing the gene's product - the RNA molecule - from the TSS where transcription begins. CAGEscan presents an instrumental tool for exploring the "RNA continent", thousands of RNAs that do not code for proteins but function to modulate the cell's genetic program. While presenting promising targets for new therapeutic approaches, these RNA have remained obscure in the past due to the limitations of existing technologies.
The two new methods greatly enhance the ability of researchers to work with tiny samples and improve the efficiency of RNA identification. Simple to perform and requiring no specialized equipment, nanoCAGE will be the technology of choice for molecular biologists, while CAGEscan will accelerate research in domains where our current knowledge of active genes is limited. Together, the two methods set the stage for new applications in drug screening, biopsy analysis and whole-transcriptome association studies.
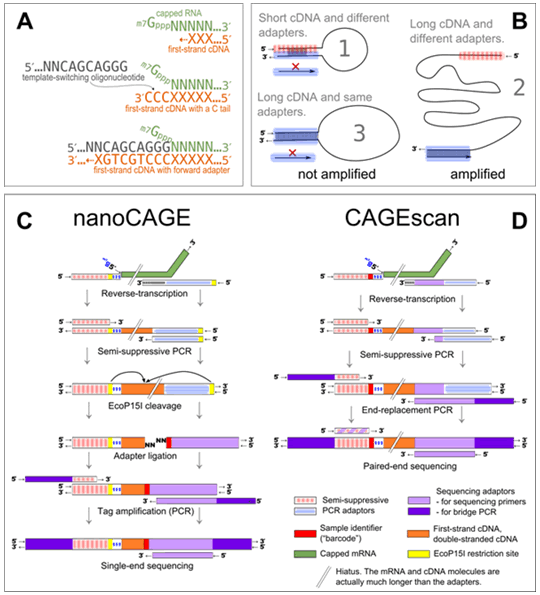
Figure 1: Experimental outline of the nanoCAGE and CAGEscan protocols.
(a) nanoCAGE captures the 5' ends of molecules by template switching. When polymerizing the cDNA of a capped mRNA, the reverse transcriptase adds extra cytosines that are complementary to the cap. Each 5' full-length cDNAs is extended upon hybridization of the riboguanosine-tailed template-switching oligonucleotides to these extra cytosines. (b) In the semisuppressive PCR, the short templates fold intramolecularly and prevent the binding of primers which precludes amplification; longer molecules are less likely to fold and are thus amplified. Templates derived from reaction artifacts form stable homoduplexes, also precluding amplification. (c) Preparation of nanoCAGE tags. After template-switching, semisuppressive PCR and EcoP151 cleavage, 25-bp tags are ligated to barcode-containing oligonucleotide adapters. After PCR amplification, the nanoCAGE tags are sequenced by synthesis. (d) Preparation of 5'-full-length cDNA libraries for paired-end sequencing with the CAGEscan protocol. Capped mRNAs capture is similar to that described in a. The ends of the amplified cDNA constructs are replaced in a PCR with adapters for sequencing in the Illumina Genome Analyzer, which produces paired-end reads from single cDNAs. Line breaks indicate that mRNA and cDNA are much longer than adaptors.
References
- Plessy C, et al. Linking promoters to functional transcripts in small samples with nanoCAGE and CAGEscan. Nature Methods 7 528–34 (2010), doi: 10.1038/nmeth.1470
- Md Salimullah, Mizuho Sakai, Charles Plessy and Piero Carninci. NanoCAGE: a high-resolution technique to discover and interrogate cell transcriptomes. Cold Spring Harbor Protocols 2011, doi: 10.1101/pdb.prot5559
- Tang DT, Plessy C, Salimullah M, Suzuki AM, Calligaris R, Gustincich S, Carninci P. Suppression of artifacts and barcode bias in high-throughput transcriptome analyses utilizing template switching. Nucleic Acids Res. 2013 Feb 1;41(3):e44. doi: 10.1093/nar/gks1128
- Harbers M, Kato S, de Hoon M, Hayashizaki Y, Carninci P, Plessy C. Comparison of RNA- or LNA-hybrid oligonucleotides in template-switching reactions for high-speed sequencing library preparation. BMC Genomics. 2013 Sep 30;14(1):665. doi:10.1186/1471-2164-14-665
